|
ISO 17025 - Internal audit checklist defines a description information of QMS internal audit check list to comply ISO 17025 technical requirements. This draft is submitted to a parallel vote in ISO and in IEC. 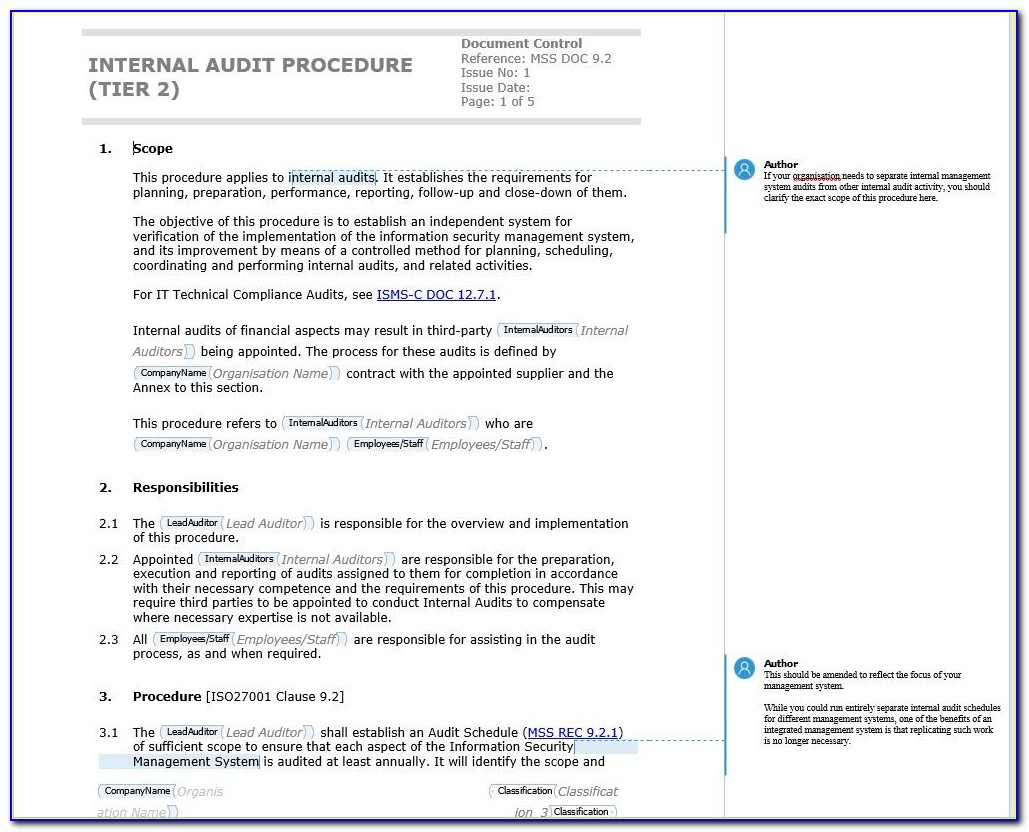
The document covered all processes are in scope. RECIPIENTS OF THIS DRAFT ARE INVITED TO SUBMIT, WITH THEIR COMMENTS, NOTIFICATION OF ANY RELEVANT PATENT RIGHTS OF WHICH THEY ARE AWARE AND TO PROVIDE SUPPORTING DOCUMENTATION. IN ADDITION TO THEIR EVALUATION AS BEING ACCEPTABLE FOR INDUSTRIAL, TECHNO LOGICAL, COMMERCIAL AND USER PURPOSES, DRAFT INTERNATIONAL STANDARDS MAY ON OCCASION HAVE TO BE CONSIDERED IN THE LIGHT OF THEIR POTENTIAL TO BECOME STAN DARDS TO WHICH REFERENCE MAY BE MADE IN NATIONAL REGULATIONS. ISO/CEN PARALLEL PROCESSING Reference number ISO/IEC FDIS 17025:2017(E) © ISO/IEC 2017ĬOPYRIGHT PROTECTED DOCUMENT © ISO/IEC 2017, Published in Switzerland All rights reserved. Unless otherwise specified, no part of this publication may be reproduced or utilized otherwise in any form or by any means, electronic or mechanical, including photocopying, or posting on the internet or an intranet, without prior written permission. ISO/IEC 17025:2017 Audit Check List lause Requirement ocument Reference Statues of the mplementation omment 6.2 Personnel 6.2.Permission can be requested from either ISO at the address below or ISO’s member body in the country of the requester. CP 401 CH-1214 Vernier, Geneva, Switzerland Tel. Global Manager Group has prepared presentation to provide information about Calibration and Testing Laboratory Accreditation Standard - ISO 17025 and about Documentation Requirements. 
5 6.3 Facilities and environmental conditions. 8 6.6 Externally provided products and services. ISO 170252017 Quality Manual Comparison - ISO 17025: 2005 Revisions Quality Manual Template The ISO 17025 Quality Manual Template is the most efficient approach for you to implement your Audit. ISO 17025 2017 INTERNAL AUDIT REPORT SAMPLE MANUALĩ 7.1 Review of requests, tenders and contracts. 9 7.2 Selection, verification and validation of methods. ISO 17025 2017 INTERNAL AUDIT REPORT SAMPLE VERIFICATIONġ0 7.2.1 Selection and verification of methods. 13 7.6 Evaluation of measurement uncertainty.ġ2 7.4 Handling of test or calibration items. 14 7.8.2 Common requirements for reports (test, calibration or sampling). This includes a discussion on how to prepare for the onsite assessment. 15 7.8.3 Specific requirements for test reports. A discussion of the process of achieving accreditation to the ISO 17025 International Standard is presented. To prepare organizations for ISO/IEC 17025 Accreditation, QSE auditors provide extensive / comprehensive ISO/IEC 17025 internal audit reports and submits them. 15 7.8.4 Specific requirements for calibration certificates. 16 7.8.5 Reporting sampling – specific requirements. 16 7.8.6 Reporting statements of conformity. 17 7.8.7 Reporting opinions and interpretations. 18 7.11 Control of data and information management. 19 Management system requirements.19 8.1 Options. 
ISO 17025 2017 INTERNAL AUDIT REPORT SAMPLE VERIFICATIONĢ0 8.2 Management system documentation (Option A).ISO 17025 2017 INTERNAL AUDIT REPORT SAMPLE MANUAL.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
